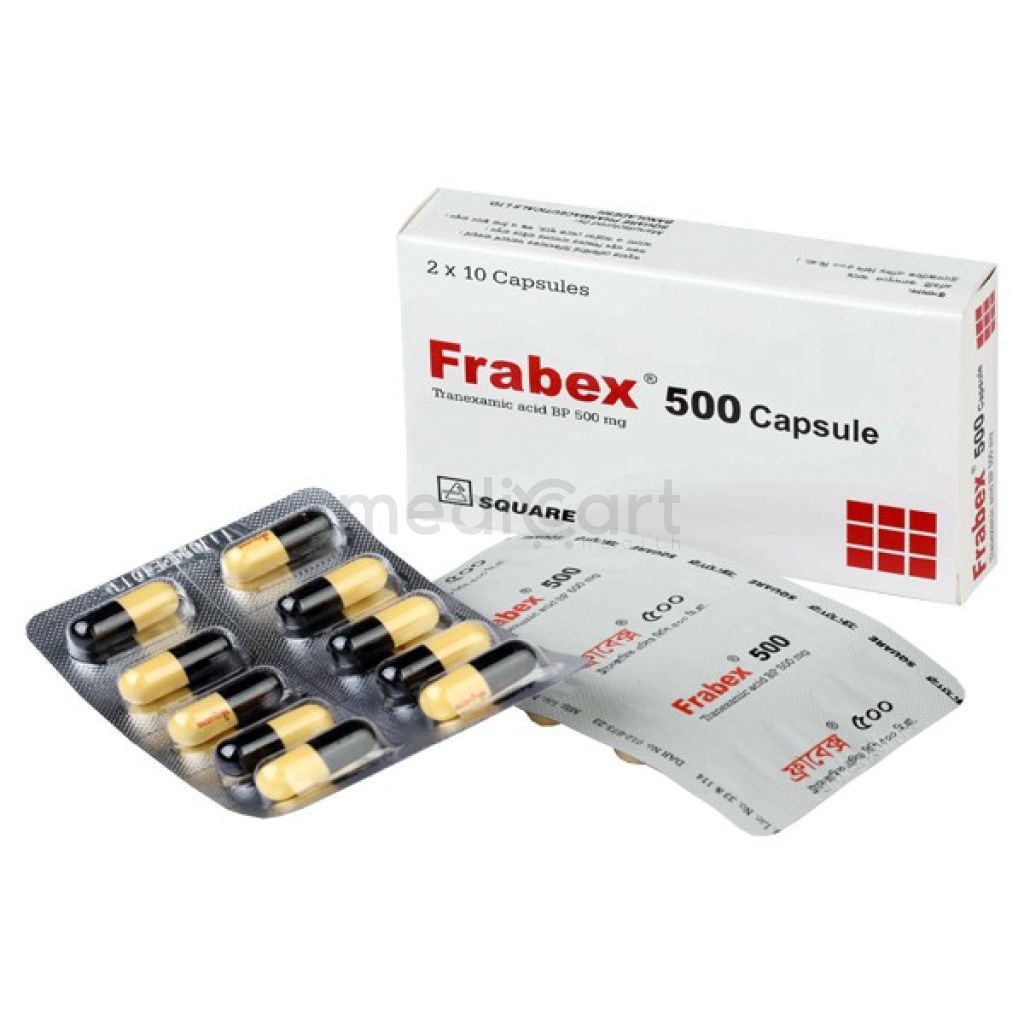

Frabex 500 mg Cap. 500mg
Capsule
Pack Size :
10 Capsule x 1 Strips
Generics :
Tranexamic Acid
Manufacturer :
Square Pharmaceuticals Ltd.
Best Price *
TK
200.00
* Delivery will be done in Dhaka city only.
Alternative Product
More Information About - Frabex 500 mg Cap. 500mg
Description
Generic Name
Tranexamic AcidPrecaution
Mild to moderate renal impairment, irregular menstrual bleeding, previous history of thromboembolic disease, haematuria. Monitor closely in disseminated intravascular coagulation. Monitor LFT and eye examination regularly during long-term use. Discontinue if disturbance in colour vision occurs. Avoid IV inj rate >1 ml/minute due to risk of hypotension. Pregnancy, lactation.Indication
Haemorrhage, Hereditary angioedema, Heavy Menstrual Bleeding (Menorrhagia), Tooth Extraction in Patients with Hemophilia, Refractory melasmaContra Indication
Severe renal failure, active intravascular clotting, thromboembolic disease, colour vision disorders, subarachnoid bleeding.Dose
N/ASide Effect
>10% Headache (50.4%),Nasal and sinus symptoms (25.4%),Back pain (20.7%),Abdominal pain (19.8%),Musculoskeletal pain (11.2%) 1-10% Arthralgia (6.9%),Muscle cramps and spasms (5.8%),Migraine (6%),Anemia (5.6%),Fatigue (5.2%) Frequency Not Defined Visual abnormalities,Hypotension (with rapid injection),Nausea,Vomiting,DiarrheaPregnancy Category
Name : B
Description
Animal reproduction studies have failed to demonstrate a risk to the fetus and there are no adequate and well-controlled studies in pregnant women OR Animal studies have shown an adverse effect, but adequate and well-controlled studies in pregnant women have failed to demonstrate a risk to the fetus in any trimester.Mode of Action
Tranexamic acid is an antifibrinolytic agent that competitively inhibits breakdown of fibrin clots. It blocks binding of plasminogen and plasmin to fibrin, thereby preventing haemostatic plug dissolution.Interaction
Potentially Fatal: Increased risk of thrombus formation with estrogens, Factor IX complex concentrates or anti-inhibitor coagulant concentrates. Increased risk of fatal thrombotic complications with tretinoin in acute promyelocytic leukaemia.Pregnancy Category Note
Pregnancy Category: B Lactation: Excreted in breast milk, not recommendedAdult Dose
Oral Short-term management of haemorrhage Adult: 1-1.5 g or 15-25 mg/kg bid or tid. Management of hereditary angioedema Adult: 1-1.5 g bid or tid. Menorrhagia Adult: 1 g tid during menstruation as necessary for up to 5 days, may be increased for heavy bleeding. Max: 4 g daily. Patients with haemophilia undergoing dental extraction Adult: 1.5 g (or 25 mg/kg) 8 hourly. Intravenous Short-term management of haemorrhage Adult: 0.5-1 g or 10 mg/kg tid or 25-50 mg/kg daily by continuous infusion.Child Dose
Oral Short-term management of haemorrhage Child: 25 mg/kg bid or tid. Management of hereditary angioedema Child: 25 mg/kg bid or tid. Intravenous Short-term management of haemorrhage Child: 10 mg/kg bid or tid.Renal Dose
Renal impairment: Adjust dose based on serum creatinine concentration: 120-250 micromol/l: 15 mg/kg bid daily; 250-500 micromol/l: 15 mg/kg once daily; >500 micromol/l: 7.5 mg/kg once daily or 15 mg/kg once every 48 hr.Administration
May be taken with or without food.Disclaimer
The information provided herein are for informational purposes only and not intended to be a substitute for professional medical advice, diagnosis, or treatment. Please note that this information should not be treated as a replacement for physical medical consultation or advice. Great effort has been placed to provide accurate and comprehensive data. However, Medicart along with its authors and editors make no representations or warranties and specifically disclaim all liability for any medical information provided on the site. The absence of any information and/or warning to any drug shall not be considered and assumed as an implied assurance of the Company.