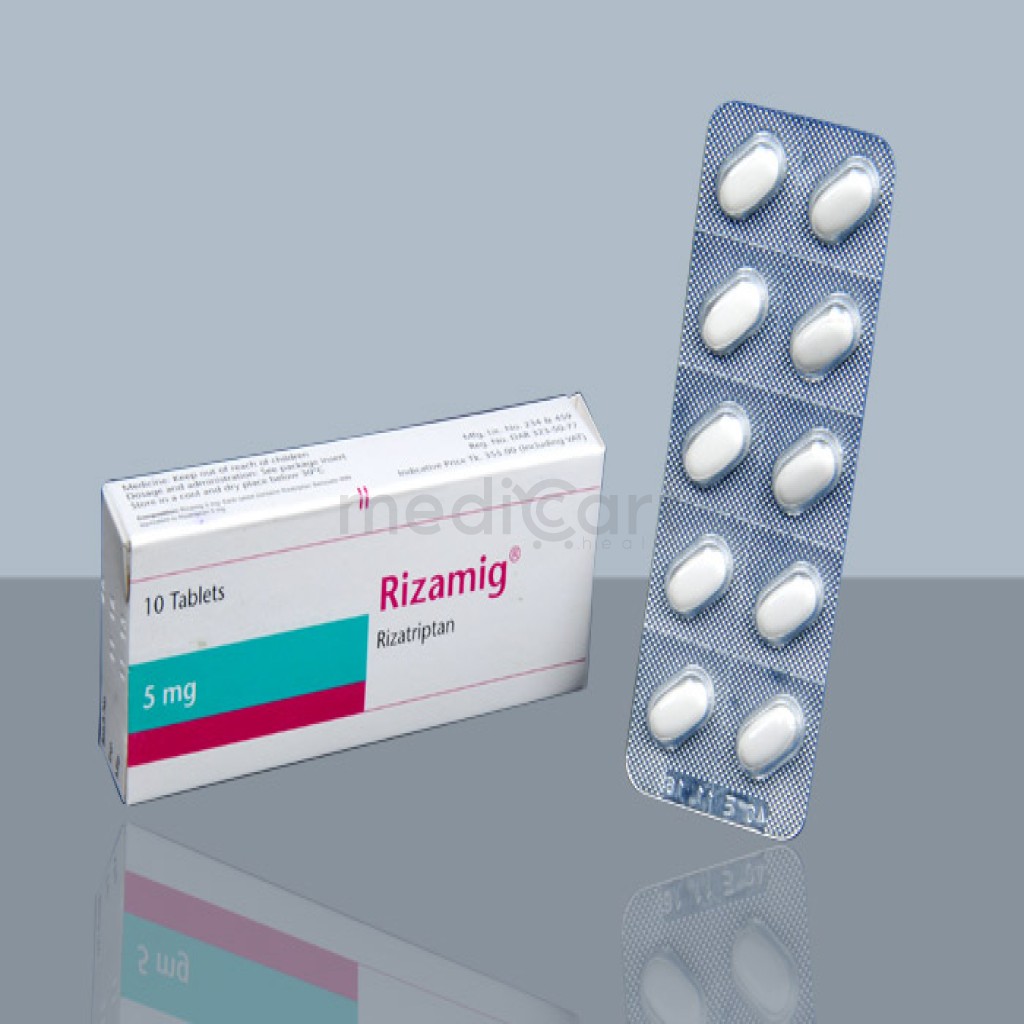

Rizamig - 5mg
Tablet* Delivery will be done in Dhaka city only.
Alternative Product
More Information About - Rizamig - 5mg
Description
Generic Name
Rizatriptan
Precaution
Elderly; mild to moderate hepatic or renal impairment; coronary artery disease; pregnancy, lactation. May cause drowsiness. History of seizures. Ensure an interval of at least 24 hr after stopping an ergotamine compound and starting a serotonin (5-HT1) agonist. Lactation: Unknown; use with caution in breastfeeding
Indication
Acute migraine attacks
Contra Indication
History of MI, peripheral vascular disease, transient ishaemic attack, ischaemic heart disease or Prinzmetal's angina; uncontrolled hypertension; basilar or hemiplegic migraine; severe hepatic or renal impairment. Adolescent <18 yr.
Dose
N/A
Side Effect
>10% Drowsiness (13-30%, dose related),Fatigue (13-30%, dose related),Dizziness (11-15%) 1-10% Dizziness (4-9%),Somnolence (4-8%),Fatigue (4-7%; dose related),Nausea (4-6%),Asthenia (1-5%),Hot flashes (1-5%),Paresthesia (3-4%),Dry mouth (3%),Chest pain (2-3%),Pain/pressure in chest, neck, throat, jaw (<2%),Headache (<2%),Dyspnea (>1%),Hypoesthesia (>1%),Palpations (>1%),Skin flushing (>1%) <1% Tachycardia,Angioedema,Wheezing,Hypertensive crisis,Bradycardia,Hallucination,Epidermal necrolysis,Hearing impairment,Arrhythmias,Myocardial infarction and coronary artery vasospasm in patients with CAD risk factors Potentially Fatal: Toxic epidermal necrolysis.
Pregnancy Category
Name : Not Classified
Description
FDA has not yet classified the drug into a specified pregnancy category.Mode of Action
Rizatriptan is a selective serotonin (5-HT1) agonist in cranial arteries responsible for vasoconstriction and reduction of inflammation associated with antidromic neuronal transmission.
Interaction
Increased serum concentrations with propranolol. Increased risk of vasospastic reactions when used with ergotamine and methysergide. Concurrent use with SSRIs may increase risk of serotonin syndrome. Potentially Fatal: Concurrent use with or within 2 wk of stopping MAOI treatment.
Pregnancy Category Note
Pregnancy Available data in pregnant women are not sufficient to draw conclusions about drug-associated risk for major birth defects and miscarriage The Pregnancy Registry for rizatriptan did not identify any pattern of congenital anomalies or other adverse birth outcomes over the period of 1998-2018; however, the lack of identification of any pattern should be viewed with caution, as the number of prospective reports with outcome information was low and did not provide sufficient power to detect an increased risk of individual birth defects associated with the use Additionally, there was significant loss to follow-up in the prospective pregnancy reports, further complicating this assessment of an association between rizatriptan and any pattern of congenital anomalies or other adverse birth outcomes Animal data Developmental toxicity was observed following oral administration of rizatriptan during pregnancy (decreased fetal body weight in rats) or throughout pregnancy and lactation (increased mortality, decreased body weight, and neurobehavioral impairment in rat offspring) at maternal plasma exposures greater than that expected at therapeutic doses in humans Clinical considerations In women with migraine, there is an increased risk of adverse perinatal outcomes in the mother, including pre-eclampsia and gestational hypertension Lactation Data are not available on the presence of rizatriptan or any active metabolites in human milk, or on the effects of rizatriptan on the breastfed infant, or on milk production Rizatriptan was excreted in rat milk, with levels in milk ~6 times those in maternal plasma
Adult Dose
Oral Acute migraine attacks Adult: Initially, 10 mg. If ineffective, 2nd dose should not be taken for the same attack. If symptoms recur after initial response, a further dose of 10 mg may be given. Doses should be separated by at least 2 hr. Max: 20 mg/24 hr. If patient is also taking propanolol, initiate with 5 mg. Max: 10 mg/24 hr. Ensure that the 2 drugs are separated by at least 2 hr. Hepatic Impairment For mild-moderate impairment, initate with 5 mg. Further dose of 5 mg may be taken after an interval of at least 2 hr. Max: 10 mg/24 hr. Avoid in severe impairment.
Child Dose
Migraine Headache Acute treatment of migraine with or without aura <6 years: Safety and efficacy not established 6-17 years (<40 kg): 5 mg PO once q24hr 6-17 years: (40 kg or greater): 10 mg PO once q24hr Dosing considerations Efficacy and safety of treatment with more than 1 dose within 24 hours in pediatric patients has not been established 6-17 years (<40 kg) taking propranolol: Do not administer rizatriptan 6-17 years (>40 kg) taking propranolol: Limit dose to 5 mg PO once q24hr as necessary; not to exceed 5 mg/24hr
Renal Dose
Renal Impairment For mild-moderate impairment, initate with 5 mg. Further dose of 5 mg may be taken after an interval of at least 2 hr. Max: 10 mg/24 hr. Avoid in severe impairment.
Administration
May be taken with or without food.
Disclaimer
The information provided herein are for informational purposes only and not intended to be a substitute for professional medical advice, diagnosis, or treatment. Please note that this information should not be treated as a replacement for physical medical consultation or advice. Great effort has been placed to provide accurate and comprehensive data. However, Medicart along with its authors and editors make no representations or warranties and specifically disclaim all liability for any medical information provided on the site. The absence of any information and/or warning to any drug shall not be considered and assumed as an implied assurance of the Company.