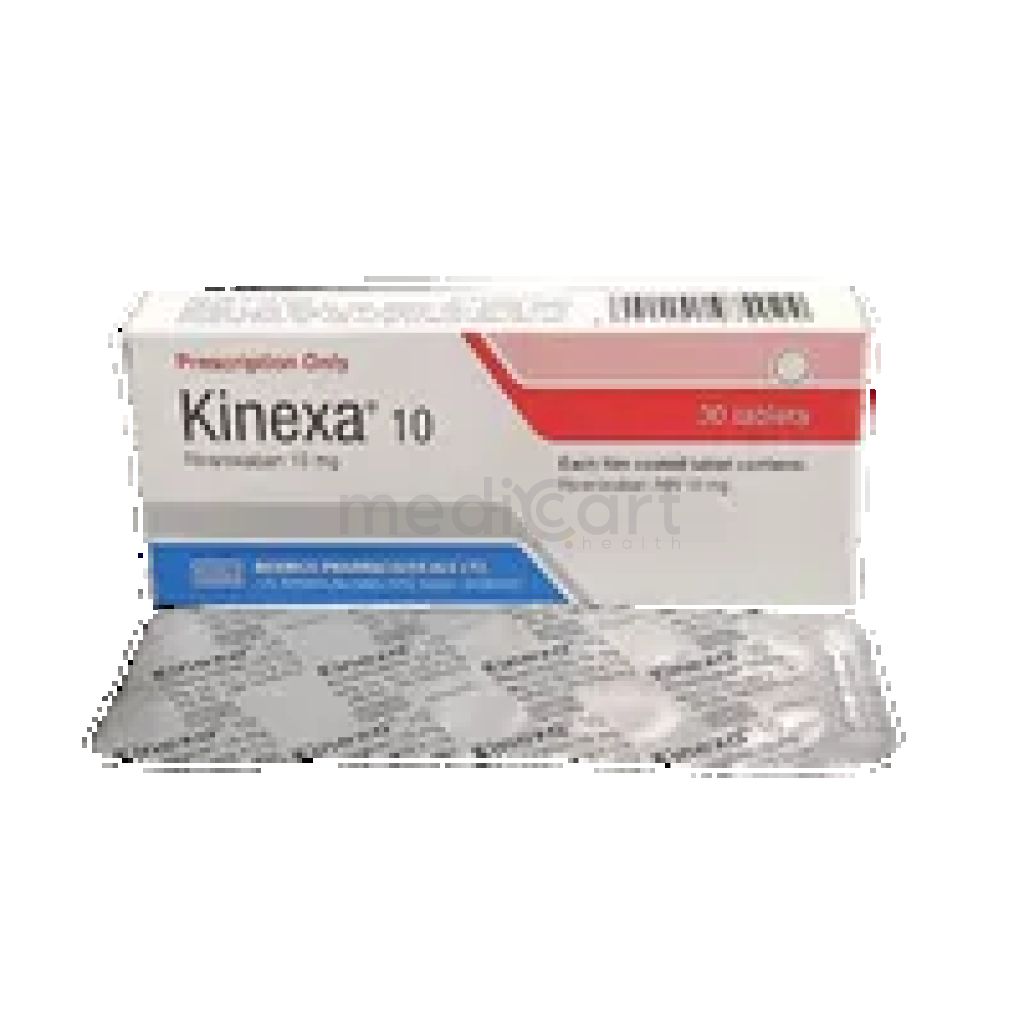

Kinexa 10 mg Tab. - 10 mg
Tablet
Pack Size :
10 Tablet x 1 Strip
Generics :
Rivaroxaban
Manufacturer :
Beximco Pharmaceuticals Ltd.
Best Price *
TK
250.00
* Delivery will be done in Dhaka city only.
Alternative Product
More Information About - Kinexa 10 mg Tab. - 10 mg
Description
Generic Name
RivaroxabanPrecaution
Patient w/ hepatic disease associated w/ coagulopathy, severe HTN, prosthetic heart valve, vascular retinopathy, bronchiectasis. Anaesth w/ post-op indwelling epidural catheter. Hepatic and renal impairment. Pregnancy and lactation. Lactation: Unknown whether distributed in human breast milk; not recommended; a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the motherIndication
Atrial fibrillation, Strokes, Deep venous thrombosisContra Indication
Patient w/ active pathologic bleeding, significant risk of major bleeding (e.g. recent GI ulcer, oesophageal varices, recent brain, spine, or ophth surgery, recent intracranial haemorrhage, malignant neoplasms, vascular aneurysm).Dose
N/ASide Effect
1-10% Abdominal pain (<2%),Back pain (<4%),Blister (1%),Bruising (3%),Constipation (<3%),Diarrhea (<5%),Dizziness (<6%),Dyspepsia (<2%),Epistaxis (4-10%),Fatigue (<3%),Headache (3-5%),Nausea (1-3%),Hematuria (<4%),Muscle spasm (1%),Oropharyngeal pain (<1%),Osteoarthritis (<2%),Peripheral edema (<6%),Pruritus (<2%),Pyrexia (1-3%),Rash (2%),Syncope (<2%),Toothache (<1%),Urinary tract infection (<1%),Vomiting (<2%),Wound secretion (<3%) Bleeding Atrial fibrillation (21%; major bleeding 6%) DVT prophylaxis (5-6%; major bleeding <1%) DVT treatment (6-10%; major bleeding 1%) Hematoma (<3%) <1% Agranulocytosis,Hepatitis,Dysuria,Fatal bleeding,Hematoma,Hemiparesis,Hemorrhage,Hypotension,Increased amylase,Increased BUN,Jaundice,Menorrhagia,Retroperitoneal bleeding,Stevens-Johnson syndrome,Thrombocytopenia,Urticaria,XerostomiaPregnancy Category
Name : Not Classified
Description
FDA has not yet classified the drug into a specified pregnancy category.Mode of Action
Factor Xa inhibitor that inhibits platelet activation by selectively blocking the active site of factor Xa without requiring a cofactor (eg, antithrombin III) for activity. Blood coagulation cascade is dependent on the activation of factor X to factor Xa via the intrinsic and extrinsic pathways plays a central role in the blood coagulation cascade. Dose-dependent inhibition of factor Xa activity observed; antifactor Xa activity is also influenced by rivaroxaban; prolongs PT and aPTT and HepTest .Interaction
Reduced hepatic & renal clearance w/ strong CYP3A4 inhibitors (clarithromycin, erythromycin, fluconazole). Increased bleeding risk w/ azole antimycotics (eg ketoconazole, itraconazole, voriconazole, posaconazole) or HIV-PIs (eg ritonavir); other anticoagulants; NSAIDs (including acetylsalicylic acid) & platelet aggregation inhibitors. Reduced plasma conc w/ CYP3A4 inducers (eg rifampicin, phenytoin, carbamazepine, phenobarb, St. John's wort). 20-mg FC tab: Increased prothrombin time/INR of warfarin.Pregnancy Category Note
Pregnancy Limited available data in pregnant women are insufficient to inform a drug-associated risk of adverse developmental outcomes Use with caution in pregnant patients because of the potential for pregnancy related hemorrhage and/or emergent delivery; anticoagulant effect cannot be reliably monitored with standard laboratory testing Consider benefits and risks for the mother and possible risks to the fetus when prescribing to a pregnant woman Females of reproductive potential requiring anticoagulation should discuss pregnancy planning Clinical considerations Pregnancy is a risk factor for VTE and risk increases in women with inherited or acquired thrombophilias; pregnant women with thromboembolic disease have an increased risk of maternal complications (eg, pre-eclampsia); maternal thromboembolic disease increases the risk for intrauterine growth restriction, placental abruption, and early and late pregnancy loss Based on pharmacologic activity of Factor Xa inhibitors and the potential to cross the placenta, bleeding may occur at any site in the fetus and/or neonate All patients receiving anticoagulants, including pregnant women, are at risk for bleeding and this risk may be increased during labor or delivery; risk of bleeding should be balanced with the risk of thrombotic events when considering the use in this setting Lactation Drug detected in human milk Insufficient data available to determine effects on breastfed child or on milk production; drug and/or its metabolites were present in milk of rats Consider developmental and health benefits of breastfeeding along with mother?s clinical need for therapy and any potential adverse effects on breastfed infant from therapy or from underlying maternal conditionAdult Dose
DVT Prophylaxis (Orthopedic Surgery) Indicated for prophylaxis of deep vein thrombosis (DVT), which may lead to pulmonary embolism (PE) in patients undergoing knee or hip replacement surgery Knee replacement: 10 mg PO qDay for 12 days; may take with or without food Hip replacement: 10 mg PO qDay for 35 days; may take with or without food Administer initial dose at least 6-10 hr after surgery once hemostasis has been established Nonvalvular Atrial Fibrillation Indicated to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation 20 mg/day PO with the evening meal DVT or PE Treatment Indicated for treatment of DVT and PE 15 mg PO q12hr for 21 days with food, THEN 20 mg PO qDay for 6 months Reduce risk for recurrent DVT or PE Indicated to reduce the risk of recurrence of DVT and PE following initial 6 months treatment for DVT and/or PE 20 mg PO qDay following initial 6 months of treatment for DVT and/or PE Hepatic impairment Moderate impairment: Not studied Avoid use in patients with moderate-to-severe impairment (Child-Pugh B) or severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathyChild Dose
Safety and efficacy not establishedRenal Dose
Renal impairment (nonvalvular AF) CrCl 30-50 mL/min: 20 mg/day CrCl 15-50 mL/min: 15 mg/day ESRD on intermittent renal dialysis: 15 mg/day Renal impairment (postoperative thromboprophylaxis) CrCl >50 mL/min: Dose adjustment not necessary CrCl 30-50 mL/min: Use with caution; dose adjustment not necessary CrCl <30 mL/min: Avoid useAdministration
10-Mg Tab: May be taken with or without food. 15- & 20-Mg Tab: Should be taken with food.Disclaimer
The information provided herein are for informational purposes only and not intended to be a substitute for professional medical advice, diagnosis, or treatment. Please note that this information should not be treated as a replacement for physical medical consultation or advice. Great effort has been placed to provide accurate and comprehensive data. However, Medicart along with its authors and editors make no representations or warranties and specifically disclaim all liability for any medical information provided on the site. The absence of any information and/or warning to any drug shall not be considered and assumed as an implied assurance of the Company.